 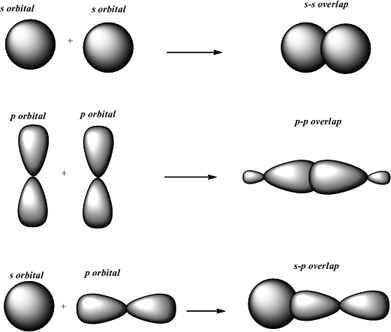
In 1s, you had two electrons,Īnd then instead of two s's, you had two electrons and onĮach of the p's, you had one, the s's and the p's all got And then instead of having 2s2Īnd then 2p2, what you essentially have is- let me And when we drew its electronĬonfiguration, in order for this to happen, carbon'sĮlectron configuration when bonding in methane needed Sp3 hybridized orbitals around the carbon, and then they eachįormed sigma bonds with each of the hydrogens. The plane of the page, and then maybe that hydrogen isīehind it, and then you have one hydrogen popping up. Tetrahedral structure, it might look like this. It kind of looks a little three-dimensional with a Hydrogens, and if I actually wanted to draw it in a way that Methane, which is literally just a carbon bonded to four So let me just make it veryĬlear what's going on here. Of conceptualize what the orbitals around the carbon Look like, or our best visual, or our best ability to kind So you have C double-bonded toĬ, and then each of those guys have two hydrogens. Visualization of how that might work, let's thinkĪbout ethene. Start forming double or triple bonds on top of a sigma bond. Than pi bonds pi bonds come into play once you What form when you have a single bond, these are stronger And it's called a pi bondīecause it's the Greek letter for essentially p, and we'reĭealing with p orbitals overlapping. Literally, with the Greek letter pi: pi bond. Top lobe here and in this bottom lobe here. Other, and you see that they overlap on this kind of Let me draw that other oneĪ little bit straighter. Has a p orbital that is parallel to this p orbital, I want to draw it a little bitīigger than that, and you'll see why a second. Let me draw them a littleīit closer together. Nucleus and I'll just draw their p orbitals. Two atoms, and I'll just draw one of each of their Kind of in the direction that they're pointing? And the other type of bond youĬould have, you can imagine if you have two p orbitals. Kind of bond could there be where my two orbitals overlap So this right here- let me make this clear. There be any other type of bond than that? Well, the other type of bond, There's an overlap kind of in the direction in which the That's the small lobe,Īnd then that's the big lobe like that. Hybridized orbital, and that's on this atom and this is kind of Me draw two nucleuses and let me just draw one When we draw tetrahedral geometries of sp3 carbons like that found in methane it is conventional to draw two bonds in the plane of the page (straight or solid lines), one bond behind the plane as a dashed line, and the fourth bond as a shaded triangle coming out of the plane. Tetrahedral geometry creates a tetrahedron which is a four-faced triangular pyramid with bond angles of 109.5 degrees between each of the hydrogens. To minimize these repulsions between the hydrogens the methane adopts a tetrahedral geometry. These hydrogen atoms each have electron clouds around them which are negative and repel each other. An sp3 hybridized carbon like methane has four bonds each going to a single hydrogen atom. And a dashed line means a bond going away from you into the plane of the page. A shaded triangle (or wedge) means a bond coming toward you out the plane of the page. A straight line (or solid line) represents a bond that is part of the plane of the page. It's meant to show the 3-D shape of bonds in molecules like the sp3 hybridized bonds in methane. They can overlap, but in different ways, and the bonds thus formed are not called sigma bonds but pi bonds. Since the other orbitals are not oriented along the bond axis, they cannot overlap "head on". pz orbital, along the z axis, and any s orbital (which is spherical) can overlap to form a sigma bond. If the z-axis is taken as the bond axis, only orbitals with the central axis along z-axis can form sigma bond. Head on overlap is actually a layman's term to specify the requirement of specific symmetries in combining atomic orbitals. A sigma bond involves head on overlap of atomic orbitals. The second question can be much more satisfactorily answered. In that concept, there is no explanation as to why we do not include the inner orbitals, but by not including them we get the right answers, and hence that became a so called "rule" of hybrid orbitals. Actually, there are no hybrid orbitals and hybridisation concept, introduced by Pauling is now obsolete and replaced with the superior molecular orbital model, which answers all the shortcomings of hybridisation, one of which you just mentioned. A god question, but unfortunately no simple answer. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
